
Premise 1: Evolution VS The Law of Conservation Of Mass
True science has always been founded on certain verifiable truths or axioms that are free from personal interpretation. These axioms have been empirically tested and repeated to the point of certainty and are called the Laws of Science. These Laws comprise the Mount Rushmore of verified scientific truth.
sci·en·tif·ic law
/ˌsīənˈtifik//lô/
In science, a “law” describes an observable occurrence in nature, typically expressed
as a mathematical equation, and that appears to be true under all circumstances
The chief characteristics of a Law of Science are:
- As a scientist takes care to make sure that the law applies to the scenario in question, the law will always hold true
- A Law of Science has graduated from theory, or hypothesis because it has been observed, tested and repeated throughout the history of known science and has been true without exception.
The Laws of Science are rooted in physical and biological reality—what makes them Laws is that no contradictory observations or evidence has ever been found that dispute them.
One of the first laws of physics children learn while in middle school is the Law Of Conservation of Mass discovered by Antoine Lavoisier in 1789 and they stated it like this “Matter can neither be created or destroyed”.
Wikipedia defines the Law of Conservation of Mass this way:
In physics and chemistry, the Law of Conservation of Mass states that for any system closed to all transfers of matter and energy, the mass of the system must remain constant over time, as the system’s mass cannot change, so quantity can neither be added nor be removed.
Wikipedia Conservation of Mass
Here’s the idea in plain English:
The Law of Conservation of Mass says that matter can’t just appear out of nowhere or vanish into nothing. If you have a closed system (meaning nothing goes in or out), the total amount of “stuff” (mass) stays the same. You can rearrange it, burn it, melt it, or mix it, but the total amount never increases or decreases.
Example: If you burn a piece of wood in a perfectly sealed container, the wood turns to ash, smoke, and gases. But if you weigh the whole container before and after, the total weight is exactly the same.The First Law of Thermodynamics is similar to the Law of Mass but regards energy and states that energy is neither created nor destroyed but just changes form.
If You Google “How did the Solar System form and evolve? ” you will get answers like these:
The formation and evolution of the Solar System began 4.5 billion years ago with the gravitational collapse of a small part of a giant molecular cloud. Most of the collapsing mass collected in the center, forming the Sun, while the rest flattened into a protopanetary disk out of which the planets, moons, asteroids, and other small Solar System bodies formed.
Wikipedia
An extremely dense point exploded with unimaginable force, creating matter and propelling it outward to make the billions of galaxies of our vast universe. Astrophysicists dub this titanic explosion the Big Bang.
Exploratorium.edu
Most of the hydrogen and helium in the Universe were created in the moments after the Big Bang. Heavier elements came later. The explosive power of supernovae creates and dispersed a wide range of elements.
NASA.gov
The Theory of Evolution and the Big Bang theory both attempt to explain the origins of life and the universe through purely natural processes, deliberately excluding the need for a Creator. The Big Bang theory proposes that all the matter, energy, space, and even time itself were once compressed into a singular, infinitely dense point—smaller than a dot on a page. This point, often referred to as a singularity, is said to have rapidly expanded in an event known as the Big Bang, giving rise to the observable universe.
However, while the Big Bang theory attempts to describe the development of the universe from that initial expansion, it fails to account for the origin of the singularity itself. It does not explain where matter, energy, or governing laws came from. The theory begins with something—an immensely dense “seed” of everything—but does not scientifically explain how or why that something existed in the first place.
The Big Bang Theory Proposes That:
- The entire universe—including all mass, energy, space, and time—came into existence from a singularity, or potentially from “nothing.”
- Prior to the Big Bang, there was no space, no time, and no physical law—not even the conservation laws themselves.
How The Big Bang Theory Appears To Conflict With The Law of Conservation
| Law of Conservation | Big Bang Theory Claim |
|---|---|
| Matter and energy do not naturally come from nothing | The universe’s matter and energy suddenly came into existence at the Big Bang |
| Conservation laws normally work within an already existing system | The universe itself is said to be the beginning of all physical reality |
| Conservation depends on time, space, and physical laws already existing | Time and space themselves are believed to have begun at the Big Bang |
| In normal science, something cannot create itself | The universe is often described as originating without a prior physical cause |
| The law describes how matter and energy behave today | The Big Bang describes a unique beginning event outside ordinary observable conditions |
The Law of Conservation of Mass states unequivocally that: “Mass and energy cannot be created or destroyed—only transformed from one form to another.” This applies to closed systems, meaning within the universe as it currently exists. All observed processes in nature confirm that something cannot come from absolutely nothing.
In 2004, over 400 scientists and researchers signed the “Open Letter on Cosmology” (sometimes called the “Cosmology Statement”), publicly stating that they had serious doubts about the Big Bang as the best explanation of the universe’s origins. This was published in New Scientist magazine. Here are just a few:
Quotes From Scientist Who Oppose The Big Bang Theory
| Name | Credential / Context | Quote |
|---|---|---|
| Geoffrey Burbidge | Astrophysicist, co-author of the B²FH paper, advocate of quasi-steady-state / steady-state cosmology | “Big bang cosmology is probably as widely believed as has been any theory of the universe in the history of Western civilization. It rests, however, on many untested, and in some cases untestable, assumptions. Indeed, big bang cosmology has become a bandwagon of thought that reflects faith as much as objective truth.” |
| Eric Lerner | Plasma physicist, author of The Big Bang Never Happened | “Whenever and wherever you look at what the Big Bang hypothesis predicts, and you compare with observations, then in almost every single case you get a mass of contradictions.” (Transcend.org) |
| Halton Arp | Astronomer, known for work on peculiar galaxies and redshift anomalies | “… you begin asking yourself questions like, was the Big Bang really like this generally assumed or maybe started out with one body which had successive fragmentation … or maybe the universe is turning itself inside out from inside … these unsettling questions which contemporary astronomers are not prepared or willing to face … despite an enormous amount of evidence piling up in this direction.” (AIP Oral Histories) |
| John Lennox | Mathematician, Oxford University, Christian apologist | “The universe cannot have created itself out of nothing. Nothing comes from nothing. Not even quantum fluctuations.” (God and Stephen Hawking, 2011) |
| Jayant Narlikar | Cosmologist, student of Fred Hoyle, critic of Big Bang | “What is the Big Bang theory where the entire universe came out of nothing, so you are violating the law of conservation of matter and energy in a big way.” |
| Alan Guth | Theoretical Physicist, MIT, Father of Inflation Theory | “Inflation takes a very small universe and produces from it a very big universe. But inflation by itself does not explain where that very small universe came from.” |
| Paul Davies | Theoretical Physicist, cosmologist, philosopher of science | “The problem of what happened before the Big Bang is a profound one, and it is hard to see how it can ever be answered by science.” (The Mind of God, 1992) |
| Victor J. Stenger | Physicist, professor, author of The Fallacy of Fine-Tuning | “The claim that the universe began with the Big Bang has no basis in current physical and cosmological knowledge. The observations confirming the Big Bang do not rule out the possibility of a prior universe.” |
| Arthur Eddington | Astrophysicist, pioneer in stellar structure & cosmology | |
| Fred Hoyle | Astronomer, coined “Big Bang” (derisively), Steady-State advocate | “The Big Bang theory requires a recent origin of the universe that openly invites the concept of creation.” |
So if mass and energy cannot come from nothing, then the Big Bang theory (at least in its classical form) seems to violate the very physical laws it depends on to describe the universe after it begins.
If everything in the Universe came into being from non-existence, that would violate the most basic scientific principle—that you can’t get something from nothing. This contradiction is why many conclude that the universe must have had a cause beyond nature—something eternal, immaterial, and powerful enough to bring space, time, matter, and energy into being.
The beginning seems to present insuperable difficulties unless we agree to look on it as frankly supernatural.
Arthur Eddington—Astrophysicist, pioneer in stellar structure & cosmology (The Expanding Universe)
If science has shown that God does not exist, it has not been by appealing to Big Bang cosmology. The hypothesis of God’s existence and the facts of contemporary cosmology are consistent.
David Berlinski—Philosopher of Science
David Berlinski argues that the only way matter could emerge from nothing is if God brought it into being. The book of Genesis itself describes a kind of ‘big bang’: there was nothing, and then God—the ultimate cause—spoke the universe into existence. In this way, the hypothesis of God’s existence aligns with the findings of modern cosmology.
Challenge Question : If matter cannot be created or destroyed in a closed system, then where did the molecules that formed the molecular cloud—or the so-called collapsing mass described in the previous explanations—originate?
- Was it spontaneously generated from nothing?
- Was the original matter eternal?
- Was the original matter created by God?
Premise 2: Evolution VS The 2nd Law Of Thermodynamics
The 2nd Law of Thermodynamics fundamentally states that systems naturally move from order toward disorder over time. No energy conversion is 100% efficient, and everything gradually runs out of usable energy. In the simplest terms, it means that every biological or physical system begins to break down and trends downward toward decay—not upward toward greater order.
For example, when a car converts chemical energy to mechanical energy, some of the energy is lost as heat during the conversion. This heat eventually dissipates into space. While this dissipated heat is still a form of energy, it can no longer be readily recaptured and used as a functional source of energy.
Physical systems (such as the human body) need energy to maintain an organized arrangement of their molecules. When there is no input of energy (making it a “closed system”), chemical bonds may break, large molecules begin to disassemble, and material structures gradually deteriorate. Thus, as a system slowly loses energy, its molecules become more disorganized and randomly distributed. This is an effect of entropy.
en·tro·py
/ˈentrəpē/
a thermodynamic quantity representing the unavailability of a system’s thermal energy for conversion into mechanical work, often interpreted as the degree of disorder or randomness in the system.
“the second law of thermodynamics says that entropy always increases with time.
2. lack of order or predictability; gradual decline into disorder.
The 2nd Law of Thermodynamics or “Entropy” is why terrestrial bodies such as stars die, and why the biological bodies of animals, and humans do not continually evolve upward over time, but devolve and break down over time. The dominating characteristics of entropy are that disorder increases while order declines.
The human body serves as a prime example of how the 2nd Law of Thermodynamics works:
Entropy—the gradual decline into disorder according to the Second Law of Thermodynamics—begins in the human body from the very moment of conception, though its effects are not equally visible throughout all stages of life. Here’s how it unfolds:
1. From Conception (Biological Level)
- Cellular processes begin with remarkable order and energy efficiency, but cellular damage and imperfections in DNA replication begin accumulating almost immediately.
- Metabolic activity produces free radicals, which over time damage cells, even in early development.
2. During Growth (Childhood to Early Adulthood)
- While the body appears to defy entropy by growing and becoming more complex, this is only possible because of a constant input of energy (food, oxygen, etc.).
- Entropy is still present—in the form of cell turnover, wear and tear, and accumulated mutations, but it’s balanced by growth and repair mechanisms.
3. After Peak Maturity (Around Ages 25–30)
- Entropy becomes more dominant as the body’s ability to repair and regenerate declines.
- Aging begins: telomeres shorten, cells become less efficient, and systems begin to slowly break down.
4. At Death
- The body can no longer resist entropy. Biological order collapses entirely, and disintegration begins rapidly as systems fail and decomposition sets in.
What causes death in humans or any other complex organism is not merely the failure of the brain, organs, or the presence of disease—it is the entropy that begins in every cell the moment life starts. All organisms are irreducibly complex, and from the outset, their cellular systems are subject to gradual breakdown and disorder. This universal tendency toward decay is the exact opposite of what evolution requires and predicts, which is an ongoing increase in complexity and functional order over time.
Evolution Is The Polar Opposite Of The 2nd Law of Thermodynamics
The Second Law of Thermodynamics presents a serious challenge to the theory of evolution. This law states that in a closed system, entropy—a measure of disorder—tends to increase over time. In simpler terms, natural processes tend toward decay, disorder, and randomness, not toward increased complexity and order.
Evolution claims the opposite: that life has progressed from simple, disorganized molecules to highly ordered, complex organisms. According to evolutionary theory, mindless, undirected processes have somehow built the vast information systems found in DNA, the precise molecular machinery of cells, and the intricate structures of the human body. This contradicts what the Second Law predicts.
Evolution Proposes Order from Disorder
- The Second Law tells us that natural systems tend toward disorder.
- Evolution requires the opposite—that over time, simple organisms became increasingly complex and structured.
- Without intelligent guidance, this trend toward increasing order seems to defy what we consistently observe in nature.
Biological Information Requires a Source
- The genetic code within DNA contains vast amounts of highly ordered, specified information.
- The Second Law implies that information and order naturally degrade over time—not spontaneously increase.
Observed Reality Aligns with Biblical Creation
- In the real world, we see systems breaking down over time—not becoming more ordered.
- This is consistent with the biblical view of a fallen creation (Genesis 3) where decay, death, and disorder entered the world.
- Creationists believe the original creation was perfect and orderly, and the Second Law reflects the current state of a world that is wearing out (Psalm 102:25–26; Romans 8:20–22).
Evolution, the argument goes, is a decrease of entropy, because it involves things getting more organized over time, while the second law says that things get more disordered over time. So evolution violates the second law.
Robert N. Oerter; Professor of Physics & Astronomy; George Mason University
The Second Law of Thermodynamics underscores that natural processes consistently move toward disorder, decay, and the breakdown of complex systems. Against this backdrop, the idea that random, undirected processes could steadily build higher levels of complexity, structure, and information over vast periods of time runs counter to what human reason and observation tell us. Energy alone does not create order—it tends to accelerate decay unless it is carefully directed. To claim that life advanced from simple molecules to sophisticated organisms through blind chance appears inconsistent with the fundamental law of entropy and contradicts the patterns we actually see in nature.
Evolution relies on time and advantage—the 2nd Law of Thermodynamics says time naturally produces disadvantage, decay, and loss of usable energy, not increasing order and complexity.
Scientists Agreeing Entropy and Evolution Conflict
| Name | Credentials | Quote |
|---|---|---|
| Jeff Miller | PhD Biomechanical Engineering | “If evolutionist cannot use science and it’s laws to bring about the Universe, then he has, in reality given up on naturalism and become a believer in supernaturalism.” |
| Robert N. Oerter | Professor of Physics & Astronomy, George Mason University | “Evolution, the argument goes, is a decrease of entropy, because it involves things getting more organized over time, while the second law says that things get more disordered over time. So evolution violates the second law.” |
| Sir Arthur Eddington | Astrophysicist, Cambridge | “If your theory is found to be against the second law of thermodynamics I can give you no hope; there is nothing for it but to collapse in deepest humiliation.” |
| A.E. Wilder-Smith | Physical Organic Chemist | “The real problem in explaining the origin of life is not the origin of the substances but the origin of the instructions to organize them… which contradicts the Second Law of Thermodynamics if attributed to chance.” |
| Robert Gange | Physicist, Research Scientist at IBM | “According to the Second Law of Thermodynamics, energy tends to dissipate; order tends to disorder. Evolutionary theory asks us to believe the opposite: that disorder gave rise to order.” |
| John Lennox | Mathematician, Oxford University | “Evolutionary biologists sometimes invoke the Second Law of Thermodynamics to dismiss design arguments, but they fail to face up to the far greater problem: how natural processes could ever increase information without violating that same law.” |
| Hyman Levy | Mathematician & Philosopher of Science | “No tendency toward progressive development is visible in the universe. The second law of thermodynamics seems to indicate that the universe is running down into chaos rather than up into order.” |
| John C. Sanford | Geneticist, Cornell University | “Mutations and natural selection cannot generate new information. In fact, they consistently cause loss of information… This fundamental reality is fully consistent with the laws of physics, which dictate that systems decay, not build themselves up.” |
| Henry M. Morris | PhD Engineering, Va. Tech | The fact that evolution is in violation of the Second Law of Thermodynamics is the most devastating argument that can be leveled against it. |
To insist that life assembled itself upward from molecules to man in spite of the relentless law of entropy is not just scientifically questionable—it is logically inconsistent with everything we know about how the world works. If skeptics want to uphold evolution, they must first answer the glaring contradiction of how increasing order can come from a principle that guarantees disorder.
Challenge Question: If the Second Law shows that order tends to decay over time, how do we reconcile the claim that life somehow developed from simplicity to complexity against this universal principle?
Premise 3: Evolution VS The Law Of Biogenesis
The Law of Biogenesis is credited to the work of Louis Pasteur and others, who, through repeated scientific experimentation and observation, established the foundational axiom: Omne vivum ex ovo—“all life comes from life.”
The idea of spontaneous generation, which suggests that life can emerge from non-living matter, has never been observed under any scientific conditions. In every observed case, life originates only from pre-existing life. Yet evolutionary scientists continue to advocate that life began by chance through a process known as chemical evolution, where non-living chemicals supposedly gave rise to the first living cell.
However, living organisms inherit complex, specified information from their parent organisms. Contrary to evolutionary claims, scientists have never observed life arising from raw, unprogrammed matter without guidance or prior information.
Evolutionary Scientist Admit there is an Unexplainable Gap in the Theory of Evolution
| Name | Credentials | Quote |
|---|---|---|
| Andrew H. “Andy” Knoll | Fisher Professor of Natural History, Harvard University (Departments of Organismic & Evolutionary Biology, and Earth & Planetary Sciences) | “The short answer is we don’t really know how life originated…” |
| Harold Urey | Nobel Laureate in Chemistry | “All of us who study the origin of life find that the more we look into it, the more we feel it is too complex to have evolved anywhere. We believe as an article of faith that life evolved from dead matter on this planet. It is just that its complexity is so great, it is hard for us to imagine that it did.” |
| Dr. James Tour | Rice University, Department of Chemistry & NanoEngineering | “I will challenge anybody, publicly, to show me a paper where we understand in detail the mechanism by which nature could have assembled the first cell. They do not exist.” |
| James Trefil | Professor of Physics, George Mason University | “We know how life, once it began, was able to proliferate and diversify until it filled every niche on the planet. Yet one of the most obvious big questions—How did life arise from inorganic matter? Remains a great unknown.” |
| American Scientist Journal | Scientific Journal | “Yet one of the most obvious big questions—how did life arise from inorganic matter? Remains a great unknown.” |
| Freeman J. Dyson | Physicist and Mathematician | “The origin of life is the deepest mystery in the whole of science… We have no idea when and how and where this gap was crossed.” |
| Leslie E. Orgel | Origin-of-Life Chemist | “One must recognize that the problem of the origin of the RNA world is far from being solved.” |
| Donald Culross Peattie | American Naturalist & Author, trained at Harvard University, noted for popular works on natural history | “A thing is either alive or it isn’t; there is nothing that is almost alive. There is but the remotest possibility of the origin of life by spontaneous generation…” |
| Hubert P. Yockey | Theoretical Biologist; Research Scientist with the U.S. Army Signal Corps and Naval Research Laboratory | “The origin of life is unsolvable as a scientific problem.” |
| Dr. James Tour | Rice University, Department of Chemistry & NanoEngineering | “The appearance of life on earth is a mystery. Scientists are confused and they are not telling the public how confused they are.” |
| George Wald | Biologist, Nobel Prize Winner | One has to only contemplate the magnitude of this task to concede that the spontaneous generation of a living organism is impossible. |
Even the simplest cell is astonishingly complex, containing thousands of proteins, molecular machines, and information-processing systems. Every function depends on precise structure and timing. Such irreducible complexity cannot arise step-by-step from blind, unguided matter. Like a watch or computer program, the pieces must all be in place and working from the start—which strongly suggests intelligent design, not chance.
Evolutionary theory proposes that inert chemicals were somehow energized until cellular life—filled with complex information and molecular machinery—arose spontaneously. Yet despite decades of highly controlled scientific experiments, no study has ever demonstrated life emerging from non-living matter, nor has such a process ever been observed in nature.
After more than 70 years of research, scientists have produced some building blocks of life, such as amino acids and nucleotides, but never a self-replicating living system from scratch. Every experiment requires precise conditions, careful timing, and intelligent intervention to keep the reactions from breaking down—conditions random evolution itself would not provide.
At the moment scientists certainly do not know how, or even if, life originated on earth from lifeless atoms. More than 30 years of experimentation on the origin of life in the fields of chemical and molecular evolution have led to a better perception of the immensity of the problem of the origin of life on Earth rather than to its solution.
Klaus Dose—Biochemist, Institute of Biochemistry, Mainz, Germany
If Complex Life Can Emerge From Matter Randomly, Why Isn’t It Happening Now?
If complex life can truly emerge randomly, spontaneously, and without purpose, as many evolutionists and naturalists claim, then why don’t we observe it happening all the time? Why did such a profound event—the origin of life from non-life—occur only once, under unknown conditions, and never again, despite vastly more time, resources, and experimentation?

If the universe is governed purely by chance and unguided processes, we would expect life to spontaneously emerge repeatedly—in different places, under a variety of conditions. Yet, what we actually observe is:
- Life only arising once, as far as we know;
- No confirmed cases of abiogenesis today, despite decades of global research;
- No laboratory success in creating life from non-life, even with unlimited energy and intelligence applied.
Life cannot have a random beginning…The trouble is that there are about two thousand enzymes, and the chance of obtaining them all in a random trial is only one part in 10 to the 40,000 power. That is a 1 with 40,000 zeros behind it.
Fred Hoyle—Prominent English astronomer, astrophysicist, and mathematician.
This points to a deeper truth: life is not the natural result of random chemistry, but the product of intentional design, information, and purpose. The singularity of life’s origin isn’t a flaw in observation—it’s a testimony to the uniqueness and mystery of life itself, suggesting it came from a singular, intelligent cause, not a repeatable natural process.
All things were made through him, and without him was not any thing made that was made. In him was life, and the life was the light of men.”
John 1:4
Even evolutionists accept the Law of Biogenesis: life comes only from life. It is what we consistently observe every day, without exception. Yet they argue that, once in history, life somehow arose from non-life. To believe this, one must assume that the Law of Biogenesis, the First Law of Thermodynamics, and even the Law of Information—which shows that complex, coded information does not arise from chaos—were all somehow bypassed for a single moment: the origin of life.
The Bible declares that life originates from the living God, not from inert matter. It teaches that the appearance of life on Earth — and nowhere else in the universe — was a miraculous act. This explains why modern science cannot account for how life began and why its naturalistic explanations so often conflict with the very laws of science.
Challenge Question : Life coming from life is a cycle that is repeated on earth a billion times a day. Scientists using the most advanced technology have not been able to produce life from non-living matter in the laboratory. Is it therefore reasonable scientist to assertively claim that it has happened before randomly, spontaneously, and undirected?
Premise 4: Time Is The Enemy of Evolution Not It’s Accomplice
Abiogenesis—the idea that life arose spontaneously from non-living matter—faces an enormous challenge when viewed through the lens of chemistry and physics. Advocates often appeal to vast stretches of time as the “solution” to the improbability problem, arguing that given enough eons, chance will eventually produce life. But time does not rescue the scenario; it worsens it:
- Chemical Fragility: Prebiotic molecules (like amino acids, nucleotides, and sugars) are chemically unstable. Left alone, they don’t patiently wait around to assemble—they degrade, cross-react into tar-like substances, or hydrolyze in water. The longer they sit, the less viable they become as building blocks.
- Entropy’s Pressure: Natural environments drive matter toward disorder, not order. Over time, the small probability of useful reactions occurring is overwhelmed by the certainty of destructive ones.
- Random Energy: Sunlight, lightning, or geothermal heat are more likely to destroy fragile molecular intermediates as to construct them. With time, degradation dominates over organization.
In this sense, time is the enemy: the longer the window, the greater the breakdown of any potential progress. Molecules don’t just appear and hang around forever — they’re constantly breaking down or reacting with other things. The stability of chemical molecules depends on their environment — things like temperature, acidity, water, light, and nearby chemicals. For example, the building blocks of RNA last only hours to days in plain water, short protein fragments can last days to months, and sugars like ribose may only last minutes to hours before falling apart.
This means evolution proposes that complex cells formed randomly from chemicals under harsh natural conditions—something scientists have never been able to produce in decades of carefully controlled laboratory experiments.
Even when molecules do form, they face threats everywhere: water breaks apart their bonds (especially sugars and RNA), they clump together into tar-like goop instead of useful chains, sunlight can fry them, oxygen can burn them up, and heat makes everything fall apart faster.
Molecular biologists, using the most advanced technology in carefully controlled laboratories, can only assemble molecules from pre-existing materials under strict conditions and precise timing. Yet evolutionists ask us to believe that this same hyper-complex process — requiring exact sequences and coordinated steps, and perfect timing — somehow occurred on the primitive Earth by sheer accident, with no control, no guidance, and no precision at all under the most unstable conditions.
We truly don’t have a clue about how unguided chemical reactions… could ever produce the necessary molecules for life.”
James Tour—Professor of Chemistry, Materials Science, and NanoEngineering
Cell and Bacteria Lifespans And Biological Limits
The time factor is absolutely critical to the Theory of Evolution. Darwin himself emphasized that his theory rests on the idea that vast stretches of time — millions of years — were necessary for countless small variations to accumulate. Only through this slow process of micromutations, he argued, could a simple single-celled organism like bacteria to eventually evolve into the vast diversity of complex life we see today.
If complex life truly evolved from a primitive cell, that original organism would have needed to survive long enough to sustain the process. But single-celled organisms are extremely fragile and short-lived. Many bacteria live only minutes or hours before dividing or dying from stress, mutation, or environmental damage. No proto-cell could remain intact for millions of years, slowly progressing toward complex life. Instead, such organisms rapidly break down, die, and decay long before any vast chain of gradual transformations could realistically occur.
Time Obstructions To Evolution of First Cell Or Bacteria
- Rapid Turnover: Most bacteria divide in minutes or hours, and individual cells die quickly under stress or lack of resources. They do not persist for millions of years to accumulate gradual, sequential complexity.
- Replication Fidelity: Each generation must faithfully copy information. Random mutations more often degrade functional systems than build them. Over countless generations, the accumulation of destructive errors outweighs rare constructive changes.
- Sequential Synthesis Bottleneck: For abiogenesis or early evolution to succeed, you would need a long chain of improbable steps building complexity in sequence. But real cells and microbes do not remain intact for such prolonged experimentation; they die, fragment, and lose continuity long before such complexity could accumulate.
Evolutionists believe a major step toward complex life happened when one simple cell swallowed another smaller bacterium. Instead of digesting it, the smaller cell supposedly survived inside the larger one and eventually became the mitochondria—the tiny “power plants” found in all complex cells. Later, they believe another cell swallowed a photosynthetic bacterium, which supposedly became the chloroplasts found in plants and algae. According to evolutionary theory, these events helped transform simple cells into the complex cells that make up plants, animals, and humans.
However, this theory faces an obvious problem: the simple cells involved—such as bacteria and archaea—are extremely short-lived. Many survive only minutes, hours, or a few days, yet the evolutionary changes required for these supposed transformations are believed to have taken hundreds of millions to over a billion years.
Evolutionist believe the entire tree of organisms needed millions of generations, involving incredibly precise mutations, coordination, and the development of entirely new regulatory systems—all began with organisms whose individual lifespans don’t even last two days.
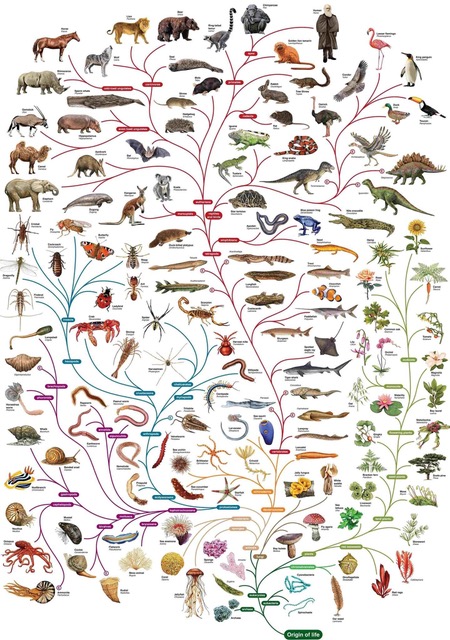
The majority of species on Earth and in evolutionary history are bacteria, insects, and other short-lived organisms. In fact the weighted average lifespan across all organisms is only 1-2 weeks.
Since 90-95% of organisms in the evolutionary tree have such short lifespans, this raises serious questions about how large-scale, information-adding evolutionary transformations could occur by chance alone.
- How can highly complex systems (e.g., organs, regulatory networks, behaviors) emerge over time when the vast majority of life reproduces quickly, mutates randomly, and dies in days?
- The average organism doesn’t live long enough to preserve major gains—or even survive long-term environmental challenges without an underlying plan or resilience mechanism.
This statistical reality reinforces the argument that unguided natural processes lack the time and biological stability necessary to build life’s complexity without intelligent design.
Challenge Question: If simple cells die in hours or days, how could blind, unguided processes slowly build the staggering complexity of life over hundreds of millions of years?
ThinkCube Truth Veracity Grid
- Have I considered the facts carefully and with an open mind?
- Is my conclusion the result of a careful examination of the facts, or is it a conclusion made in spite of the facts?
- Is my conclusion the one that makes the most sense of the evidence?
